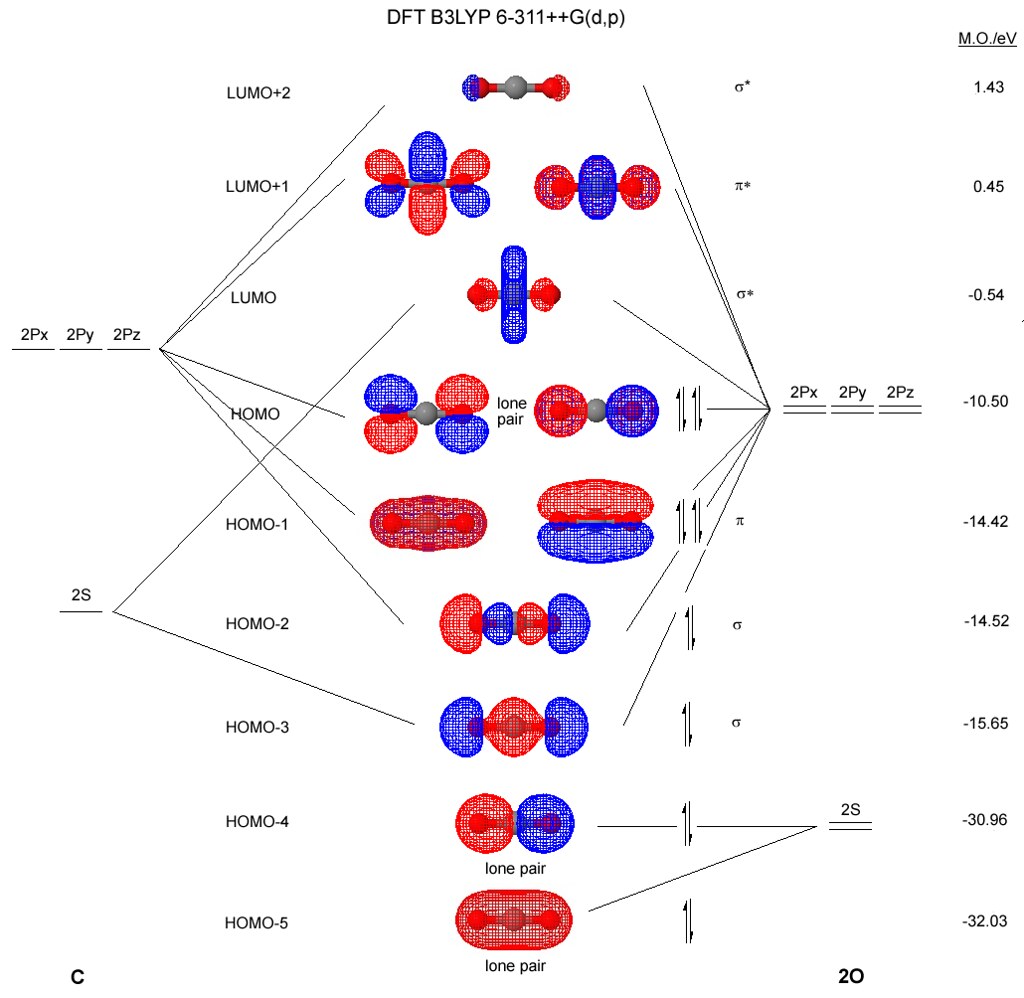
The central atom, boron, contributes three valence electrons, and each chlorine atom contributes seven valence electrons. I like the teaching method of arranging the Carbon in the center, then the oxygen atoms, then the inner bonds, the outer, and filling in the Carbon valence shell. Tetrahedral molecular structure is seen in several molecules, the most common of which is methane, CH4. Due to these repulsive forces between the valence shell electron pairs, the CO2 molecule acquires a linear shape to keep the repulsion at the least. Hence there are eight valence electrons for Carbon atoms. As we already know, the number of the attached atom to each oxygen is 2, and lone pairs on each oxygen are also two. It contains the molecules overall form, bond lengths, bond angles, torsional angles, and any other geometrical characteristics that govern each atoms position. So, the total number of unbonded pair electrons =, and the total number of bonded pair electrons =. is very simple and the procedure for drawing it same as the other molecules. From the Lewis structure, it can be seen that both the carbon atoms are associated with one another through a triple bond which is formed by one sigma () bond and 2 pi () bonds. Using chlorine dioxide minimizes the amount of toxic organochlorine compounds that are produced as byproducts. Whereas molecular geometry of H2O2 is bent because the presence of lone pair on oxygen causes the OH bond in the H2O2 lewis structure to be pushed downward and upward directions. The total valence electron in H2O2 is 14.  
PUGVIEW FETCH ERROR: 403 Forbidden National Center for Biotechnology Information 8600 Rockville Pike, Bethesda, MD, 20894 USA Contact Policies FOIA HHS Vulnerability Disclosure National Library of Medicine National Institutes of Health A difference in electronegativity among two atomic elements in the range of around 0.4 2 is considered to be polar covalent bonds. For example, Nitrogen will tend to have a Neon configuration and Chlorine will tend to have an Argon configuration. I write all the blogs after thorough research, analysis and review of the topics. It is very useful for sterilizing medical equipment. The octets of all the atoms are complete in the Lewis structure of Acetylene. Its electron pair geometry is tetrahedral. Water has 8 electrons around the central oxygen atom. Formal charge for O bonded to H and C both = 6 0.5*4 4 = 0. It considers the electron pairs (both bonded and unbonded) and talks about the concept of minimum repulsion. In order to do so, we will bring two more electrons around the atom. As we discussed H2O2 molecular shape is bent But you can find it by using the VSEPR(Valence shell electrons repulsion) theory and AXN method also. The concept of hybridization waves path for the molecular orbital diagram influencing the idea that the atomic orbitals combine and overlap to fuse and form hybrid orbitals which directly affects the molecular geometry and the bonding behavior of the newly produced molecule. Now, let us decipher the hybridization type for methanoic acid. Your email address will not be published. \) Some examples of polar molecules based on molecular geometry (HCl, NH3 and CH3Cl).CO2 Molecular Geometry The molecular Geometry of any compound is based on the arrangement of atoms, electron pairs, and bonds.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
